 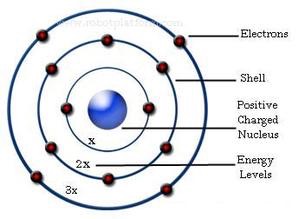 
Of all the photons (quantum packets of light energy) that an atom can absorb, only those having an energy equal to the energy difference between allowed electron orbits will be absorbed. Atoms may acquire energy that excites electrons by random thermal collisions, collisions with subatomic particles, or by absorbing a photon. Atoms with electrons in their lowest energy orbits are in a “ground ” state, and those with electrons jumped to higher energy orbits are in an “excited ” state. Subshells or suborbitals (designated s,p,d, and f ) with differing shapes and orientations allow each element a unique electron configuration.Īs electrons move farther away from the nucleus, they gain potential energy and become less stable. The first shell can hold up to two electrons, the second shell (n=2) up to eight electrons, and the third shell (n=3) up to 18 electrons. 
Increasing numbers of electrons can fit into these orbital shells according to the formula 2n 2. The orbital shells are not spaced at equal distances from the nucleus, and the radius of each shell increases rapidly as the square of n. Additional orbital shells are assigned values n=2, n=3, n=4, etc. This first orbital forms a shell around the nucleus and is assigned a principal quantum number (n) of n=1. In the Bohr model, the most stable, lowest energy level is found in the innermost orbit. To account for the observed properties of hydrogen, Bohr proposed that electrons existed only in certain orbits and that, instead of traveling between orbits, electrons made instantaneous quantum leaps or jumps between allowed orbits. In addition, physicist James Clark Maxwell ’s influential studies on electromagnetic radiation (light) predicted that an electron orbiting around the nucleus according to Newton ’s laws would continuously lose energy and eventually fall into the nucleus. Spectroscopic experiments, however, showed that hydrogen atoms produced only certain colors when heated. This predicted that when, for example, a hydrogen atom was heated, it should produce a continuous spectrum of colors as it cooled because its electron, moved away from the nucleus by the heatenergy, would gradually give up that energy as it spiraled back closer to the nucleus. The classical model of the atom allowed electrons to orbit at any distance from the nucleus. During this time Bohr developed his model of atomic structure.īefore Bohr, the classical model of the atom was similar to the Copernican model of the solar system where, just as planets orbit the sun, electrically negative electrons moved in orbits about a relatively massive, positively charged nucleus. After graduation, Bohr worked in England with Thomson and subsequently with Rutherford. While working on his doctoral dissertation at Copenhagen University, Bohr studied physicist Max Planck ’s (1858 –1947) quantum theory of radiation. 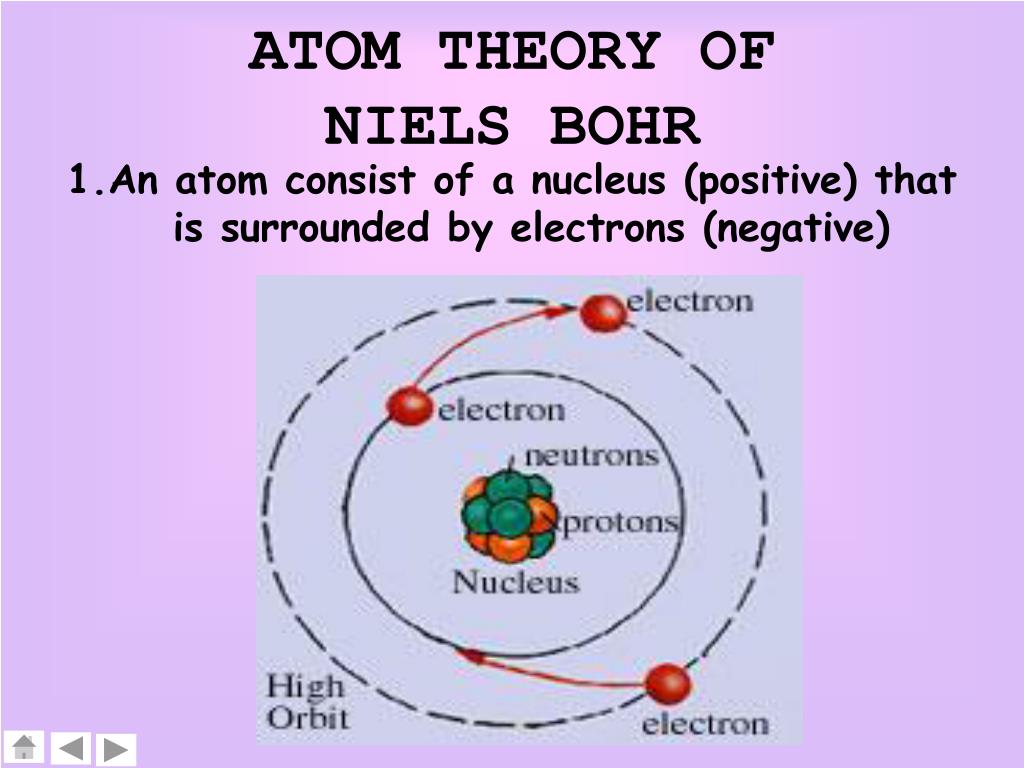
Thomson and Ernest Rutherford by incorporating quantum theory. Published in 1913, Bohr ’s model improved the classical atomic models of physicists J. However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms.The Bohr model of atomic structure was developed by Danish physicist and Nobel laureate Niels Bohr (1885 –1962). Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. (Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. \): Bohr's atomic model hydrogen emission spectra. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
